Cizumab Bevacizumab Injection
Price 7197.0 INR/ Piece
Cizumab Bevacizumab Injection Specification
- Indication
- Metastatic colorectal cancer, lung cancer, kidney cancer, glioblastoma, cervical cancer, ovarian cancer and other solid tumors
- Dosage Form
- Injection
- Salt Composition
- Bevacizumab 400 mg/16 ml
- Feature
- Monoclonal antibody; inhibits angiogenesis by targeting VEGF
- Ingredients
- Bevacizumab
- Application
- Anti-cancer therapy via intravenous infusion
- Temperature Needed For Fermentation
- 2 C to 8 C Celsius (oC)
- Physical Color/Texture
- Clear to slightly opalescent, colorless to pale brownish-yellow solution
- Fermentation Smell
- Odorless
- Storage Instructions
- Store at 2C-8C. Do not freeze. Protect from light.
- Shelf Life
- 30 months
- Marketed By
- Hetero Healthcare
- Contraindications
- Hypersensitivity to bevacizumab or any excipients
- Diluent Used
- 0.9% Sodium Chloride Injection (do not use dextrose)
- Approved Use
- For use in adults only
- Pack Size
- Single use vial of 16 ml
- Prescription Status
- Prescription only
- Concentration
- 25 mg/ml
- Molecular Type
- Recombinant humanized monoclonal antibody
- Reconstitution
- Not required, provided as ready-to-use solution
- Route of Administration
- Intravenous infusion after dilution
- Packaging Type
- Glass vial with flip-off seal
Cizumab Bevacizumab Injection Trade Information
- Minimum Order Quantity
- 2 Pieces
- Delivery Time
- 24 Hours
About Cizumab Bevacizumab Injection
Cizumab Bevacizumab Injection is a high-quality, targeted monoclonal antibody formulation used in the treatment of various types of cancers. It works by inhibiting vascular endothelial growth factor (VEGF), thereby preventing the formation of new blood vessels that tumors need to grow and spread.
This injection is widely prescribed as part of combination therapy for conditions such as metastatic colorectal cancer, non-small cell lung cancer, glioblastoma, renal cell carcinoma, and certain gynecological cancers. Manufactured under stringent quality standards, Cizumab ensures safety, efficacy, and consistent performance.
Key Features:
- Contains Bevacizumab as the active ingredient
- Anti-angiogenic action to restrict tumor growth
- Used in multiple oncology indications
- High purity and clinically proven formulation
- Manufactured in WHO-GMP compliant facilities
Packaging Details:
- Available in sterile vial form
- Different strengths available as per requirement
- Secure, tamper-proof packaging
Usage:
- Administered intravenously under the supervision of a qualified healthcare professional
- Dosage and duration depend on the patient's condition and medical history
Storage:
- Store in a refrigerator (2C to 8C)
- Do not freeze
Ready-to-Use, Highly Targeted Anti-Cancer Solution
Cizumab Bevacizumab Injection is uniquely designed as a single-use vial with a pre-prepared solution requiring no reconstitution. Its recombinant monoclonal antibody formulation specifically inhibits the growth of new blood vessels in tumors, making it effective against various cancers when prescribed by your oncologist.
Safe and Convenient Administration
Packaged in a 16 ml glass vial with a flip-off seal, Cizumab is easy to handle and intended for intravenous infusion after dilution with 0.9% sodium chloride. It must not be diluted with dextrose solutions and should only be administered by healthcare professionals.
Storage and Shelf Life Assurance
To guarantee potency and safety, store Cizumab at 2C to 8C; avoid freezing and protect from light. The product retains its efficacy for up to 30 months when stored properly.
FAQ's of Cizumab Bevacizumab Injection:
Q: How should Cizumab Bevacizumab Injection be administered?
A: Cizumab must be administered via intravenous infusion after dilution with 0.9% sodium chloride. It is not suitable for dextrose dilution, and the procedure should only be performed by qualified healthcare professionals.Q: What is the main benefit of using Cizumab for cancer therapy?
A: Cizumab works by inhibiting the formation of blood vessels (angiogenesis) in tumors, which helps to restrict tumor growth and spread, offering a targeted therapeutic approach for several types of solid tumors.Q: When is Cizumab indicated for use?
A: Cizumab is prescribed in the treatment of metastatic colorectal cancer, non-small cell lung cancer, kidney cancer, glioblastoma, cervical cancer, ovarian cancer, and other solid tumors in adults.Q: Where should Cizumab vials be stored?
A: Cizumab vials must be stored between 2C and 8C in a refrigerator; they should not be frozen and must be protected from exposure to light.Q: What process must be followed before administering Cizumab?
A: No reconstitution is required. The solution is provided ready-to-use. Before infusion, it should be diluted only with 0.9% sodium chloride, and administration must occur under medical supervision.Q: Who should avoid receiving Cizumab Bevacizumab Injection?
A: Individuals with known hypersensitivity to bevacizumab or any of the excipients in the formulation should not receive Cizumab.Q: What is the physical appearance and smell of Cizumab?
A: The solution is clear to slightly opalescent, ranging from colorless to pale brownish-yellow, and it is odorless.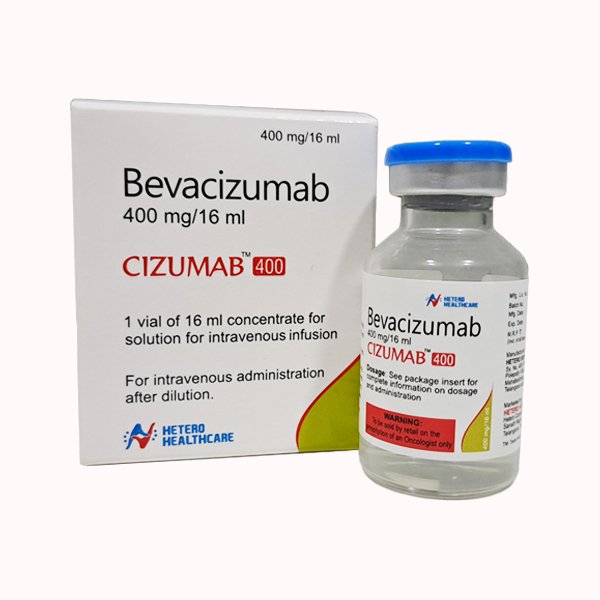

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Medicines Category
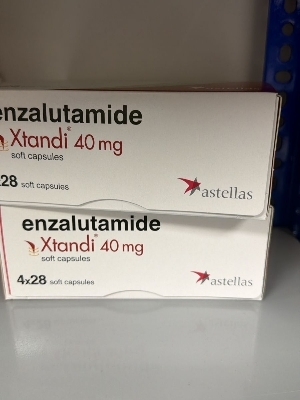
Xtandi 40mg tablet
Price 180000 INR / Tablet
Minimum Order Quantity : 1 Tablet
Physical Color/Texture : Other
Shelf Life : 12M+
Temperature Needed For Fermentation : 25
Enzyme Types : Enzyme Preparations
Heparel 200 Iu
Price 3600.0 INR / Piece
Minimum Order Quantity : 1 Piece
Physical Color/Texture : Other , Light brown powder, freeflowing
Shelf Life : 24 months from manufacturing date if stored properly
Temperature Needed For Fermentation : 80C 90C (optimum activity range)
Enzyme Types : Other, AlphaAmylase
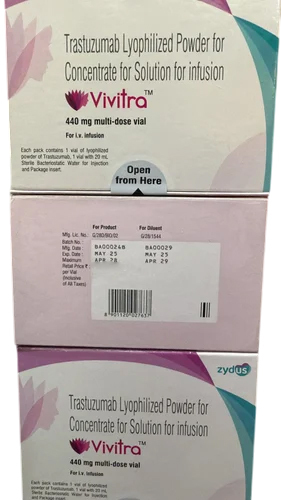
Trastuzumab Lyophilized Powder
Price 6500 INR / Piece
Minimum Order Quantity : 10 Pieces
Physical Color/Texture : Other , Powder
Shelf Life : 24 Months
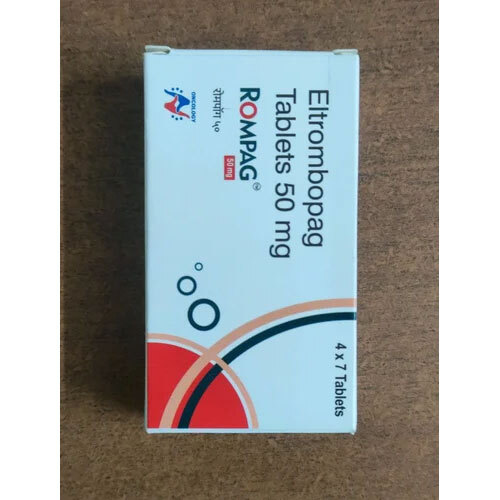
Rompag 50mg Tab
Price 550.0 INR / Piece
Minimum Order Quantity : 20 Pieces
Physical Color/Texture : Other
Shelf Life : 24 Hours
Temperature Needed For Fermentation : rankine Rankine
Enzyme Types : Emulsifiers


 Send Inquiry
Send Inquiry